
New Enhanced AO & XO Activity Test Systems Available, Major CYP Activity Not Significantly Impacted
XenoTech Establishes Method for High Aldehyde Oxidase and Xanthine Oxidase Activity in Pooled Human Liver Test Systems
Adds New In Vitro Drug Development Test Systems with Enhanced AO & XO Activity without Significantly Impacting Major CYP Activity
Today, XenoTech announced the addition of the H0606.S9(AX) Pooled Human Liver S9 and H0606.C(AX) Pooled Human Liver Cytosol test systems to the company’s catalog of in vitro drug development products. These new test systems have been prepared from tissue treated to preserve high AO (aldehyde oxidase) and XO (xanthine oxidase) activity.
Non-transplant quality human livers donated for research are a common source of both cellular and subcellular material utilized in in vitro drug metabolism studies. At the time of organ recovery, livers are flushed with an ice-cold perfusion and storage solution that preserves the tissue during delivery from the recovery site to the research facility. The two most commonly used cold storage solutions are UW (University of Wisconsin) solution and Custodiol HTK (histidine tryptophan ketoglutarate) solution.
One notable difference between these two solutions is the presence of 1 mM allopurinol in the UW solution. Researchers at Washington State University had previously shown that 1 mM allopurinol inhibits XO activity in human liver cytosol. Concerned that the exposure of human livers to allopurinol could lead to an underestimation of the contribution of XO in the metabolism of new chemical entities, XenoTech scientists also set out to evaluate XO and AO activities in S9 subcellular fractions prepared from human livers that were flushed and stored in UW solution or HTK solution. To determine whether UW or HTK affected other drug metabolizing enzymes, the metabolism of selective marker substrates for the major cytochrome P450 (CYP) enzymes was also examined.
In a poster presented in June 28, 2017, at the 14th European ISSX Meeting in Cologne, Germany, XenoTech scientists showed that pooled human liver S9 prepared from tissue that is cold-preserved in HTK solution has increased XO and AO activity with little effect on major CYP activity compared with S9 prepared from livers preserved in UW solution.
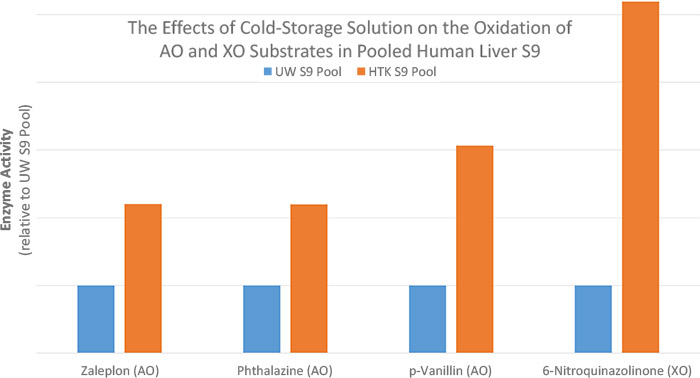
Stephanie Helmstetter, Senior Manager of Production and Quality Control at XenoTech, explained, “Aldehyde oxidase activity, as measured by the oxidation of zaleplon to 5-oxo-zaleplon, phthalazine to 1-phthalazinone, and p-vanillin to vanillic acid, was on average 2- to 3-fold higher in human liver S9 prepared from HTK-preserved livers than UW-preserved livers. Xanthine oxidase activity, as measured by the oxidation of 6-nitroquinazolinone to 6-nitroquinazolinedione, was on average over 5-fold higher in human liver S9 prepared from HTK-preserved livers than UW-preserved livers. Furthermore, preservation of livers in HTK or UW cold storage solutions did not lead to a significant difference in CYP activity.”
Zell Woodworth, Products Division Director for XenoTech, added, “Based on these results, it was clear that we needed to provide pooled human liver S9 and cytosol prepared from tissue exclusively cold-preserved in HTK solution available as a standard in vitro test system for more accurate evaluation of aldehyde oxidase and xanthine oxidase activity.”
The new H0606.S9(AX) Pooled Human Liver S9 and H0606.C(AX) Pooled Human Liver Cytosol test systems as well as other published research and in vitro drug development test systems are available on XenoTech’s website.
Other Recent Announcements from XenoTech
XenoTech’s Analytical Services Department Adds New Instrumentation
XenoTech Awarded Cryopreservation Patent, Achieves Numerous Hepatocyte Enhancements
New XenoTech VP of Scientific Operations and VP of Scientific Consulting Appointed
XenoTech Adds New Drug Transporters to Portfolio
XenoTech Adds New Dermal Subcellular Fraction Test Systems
About XenoTech
XenoTech, LLC is a global Contract Research Organization utilized by 98% of top pharma companies and numerous other organizations. For almost 25 years, the company has offered proven drug development expertise, providing an ever-evolving selection of cell and tissue-based products, screening, radiolabeling, API manufacturing, in vitro ADME Tox and pharmacology, in vivo ADME Tox and QWBA, metabolite ID and production, bioanalytical services and consulting. XenoTech’s product selection includes a wide range of high-quality unique and standard reagents, including subcellular fractions, hepatocytes, media, tissue samples, cell lines, recombinant enzymes, substrates & metabolites and more. The company also regularly prepares and delivers custom-designed products and services in response to client requests. For additional information, please refer to the company’s website at www.xenotech.com.