
XenoTech recently fulfilled its largest order of human liver subcellular fractions to date. A large pharmaceutical customer ordered over 30,000 vials of microsomes, 5,000 vials of S9 fractions and 5,000 vials of cytosol.
In order to fulfill the order, XenoTech designed an initial human liver microsome test pool from 50 individuals with optimal activities according to ranges provided by the client. Upon validation and approval of the test pool, the final pool was prepared for a total protein volume of 300,000 mg of pooled microsomes, 50,000 mg of pooled S9 fraction and 25,000 mg of pooled cytosol. XenoTech also provided 810 mg of individual donor microsomes, 250 mg of individual S9 fraction and 125 mg individual cytosol.
An array of genotype information for various metabolic enzymes of each donor was included in the pool. The human liver microsome pool was characterized according to the following assays:
- Phenacetin O-deethylation (CYP1A2)
- Coumarin 7-hydroxylation (CYP2A6)
- Bupropion hydroxylation (CYP2B6)
- Paclitaxel 6α-hydroxylation (CYP2C8)
- Diclofenac 4′-hydroxylation (CYP2C9)
- S-Mephenytoin 4′-hydroxylation (CYP2C19)
- Dextromethorphan O-demethylation (CYP2D6)
- Chlorzoxazone 6-hydroxylation (CYP2E1)
- Testosterone 6β-hydroxylation (CYP3A4/5)
- Glucuronidation of β-Estradiol (UGT1A1)
- Glucuronidation of morphine (UGT2B7)
- Benzydamine N-oxidation (FMO3)
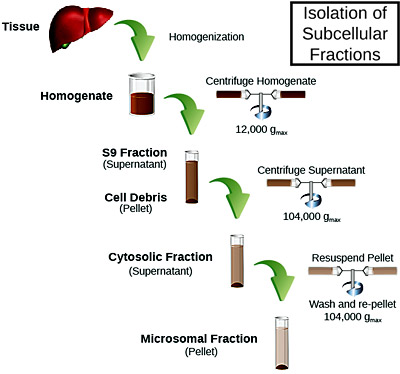
Characterization included Km/Vmax determination, and each assay was performed in duplicate on two separate days. Compiled data, including enzymatic activity and donor information tables, was then provided in both electronic and paper formats.
“This order is an excellent example of XenoTech’s capabilities,” said Jason Neat, COO of XenoTech. “Our world-renowned in vitro products and reagents facilitate research by companies worldwide and provide the foundation behind our in vitro ADME / DDI service expertise.”
Learn more about:
About the Authors
Related Posts
XenoTech Featured in Fall Issue of NewsWave
Subscribe to our Newsletter
Stay up to date with our news, events and research

Do you have a question or a request for upcoming blog content?
We love to get your feedback

