Commentary to provide context and explain significance of research is sometimes just as important as the research itself. That’s why our experts make it a priority to provide you with helpful content distilled from the many years of experience our team has accumulated. The Drug Development Insights blog contains overviews and deep dives into the drug metabolism and pharmacokinetics topics you care about—regulatory expectations, risk assessment, safety considerations, scientific innovation, study design elements, interpretation of results, and more. Explore our entries chronologically or by category to get insight into the many pieces of your drug’s development.


Drug Development Blog
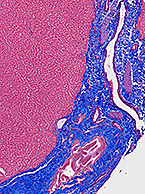
New Liver Tissue Microarrays Available – What Would Benefit Your Research?
- Disease-Specific Resources
- April 2, 2019
- Madison Esely-Kohlman, Dr. Maciej Czerwinski
As technology evolves to better meet needs in exploration of drug metabolism pathways and disease mechanisms, familiarization with appropriate and...
Disease-State Test Systems
- Test Systems & Methods
- January 13, 2019
- Michael Millhollen, Dr. Chris Bohl
XenoTech has been working with and supplying reagents for the pre-clinical ADME field for well over two decades. Years of...
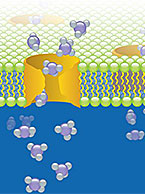
Important Considerations for the Conduct of In Vitro Drug Transporter Assays
- Drug Transporters
- January 11, 2019
- Greg Loewen, Andrea Wolff, Michael Millhollen
Not only can drug transporters affect the absorption and excretion of drugs, they can be involved in pharmacokinetic-based drug-drug interactions...
5 Keys to Set Your Contract Research Organization (CRO) Collaboration Up for Success
- Updated Offerings
- January 7, 2019
- Michael Millhollen, Kelsey Acree
Over the past 25 years of performing ADME/DMPK/DDI contract research, we’ve learned some key strategies to maximize the efficiency and effectiveness...
Holiday CSR Activities
- Updated Offerings
- December 26, 2018
- Michael Millhollen, Deja Coffin
XenoTech understands that a better future starts with our actions today, and encourages its global family to remain focused on...
FDA Guidance: Many In Vitro DDI Evaluations Should Precede FIH Studies
- Regulatory Guidance
- December 6, 2018
- Dr. Brian Ogilvie, Andrea Wolff
In October 2017, the FDA released its much-anticipated draft guidance documents for drug-drug interaction (DDI) studies, which was finalized in...
Coverage from the October 2018 PBSS DDI Workshop
- Drug Transporters
- November 5, 2018
- Greg Loewen, Michael Millhollen
The Pharmaceutical and BioScience Society (PBSS) hosted the workshop “Drug-Drug Interactions: Update on Risk Assessment, Clinical Evaluation and Regulatory Requirements” on October...
October Presentations on In Vitro Effects of Biologics on CYP Enzymes and Regulatory DDI Guidances
- Regulatory Guidance
- October 26, 2018
- Michael Millhollen
On Oct. 15th at the Peptide ADME Discussion Group Workshop in Gothenburg, Sweden, Dr. Brian Ogilvie presented on In vitro Direct and Cytokine-Mediated...
To GLP or not to GLP?
- Regulatory Guidance
- October 5, 2018
- Scott Hickman, Dr. Brian Ogilvie, Tim Patterson
That is the question. . . Knowing the answer may save you time and money Good Laboratory Practices (GLP) are...
New Recommendation: Evaluate NMEs for OCT1 Transporter-Mediated DDI Potential
- Drug Transporters
- October 1, 2018
- Dr. Brian Ogilvie, Andrea Wolff
Brian Ogilvie, Ph.D., Vice President of Scientific Consulting at XenoTech, presented a case study on the importance of the hepatic OCT1...
Subscribe to our Newsletter
Stay up to date with our news, events and research

Do you have a question or a request for upcoming blog content?
We love to get your feedback